Properties and structure of atoms
Atoms: structure and mass
Electrons and the periodic table
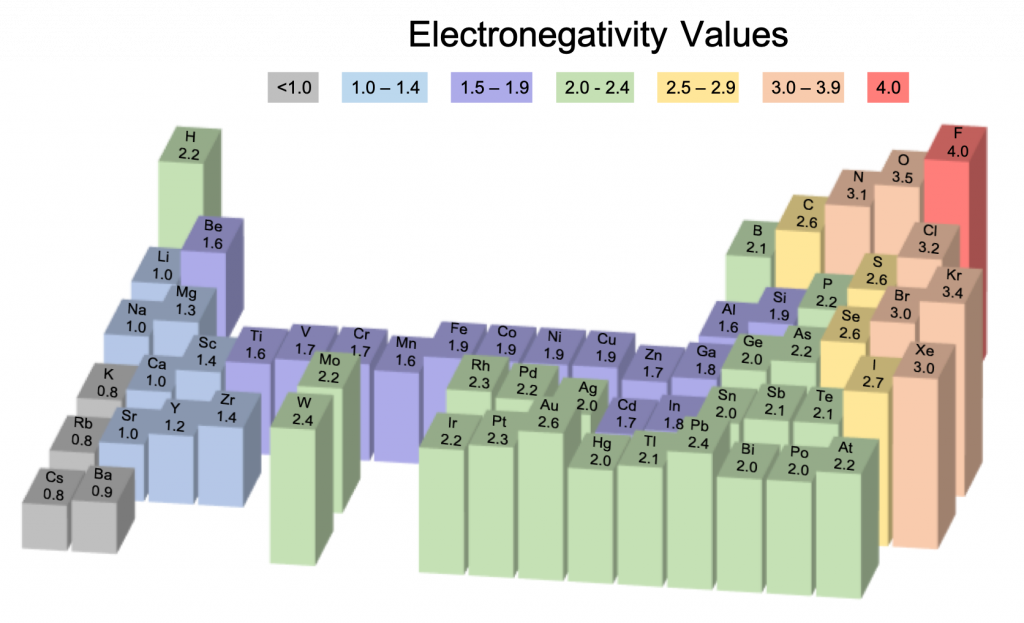
A 3D view of electronegativity values. In this view, elements without Pauling electronegativity values are excluded. Electronegativity values derived by Pauling follow predictable periodic trends with the higher electronegativities toward the upper right of the periodic table. The height of the bars indicates the electronegativity value.
Properties and structure of materials
Ionic Bonding and Materials
Useful version of the periodic table showing common ionic forms of the elements:
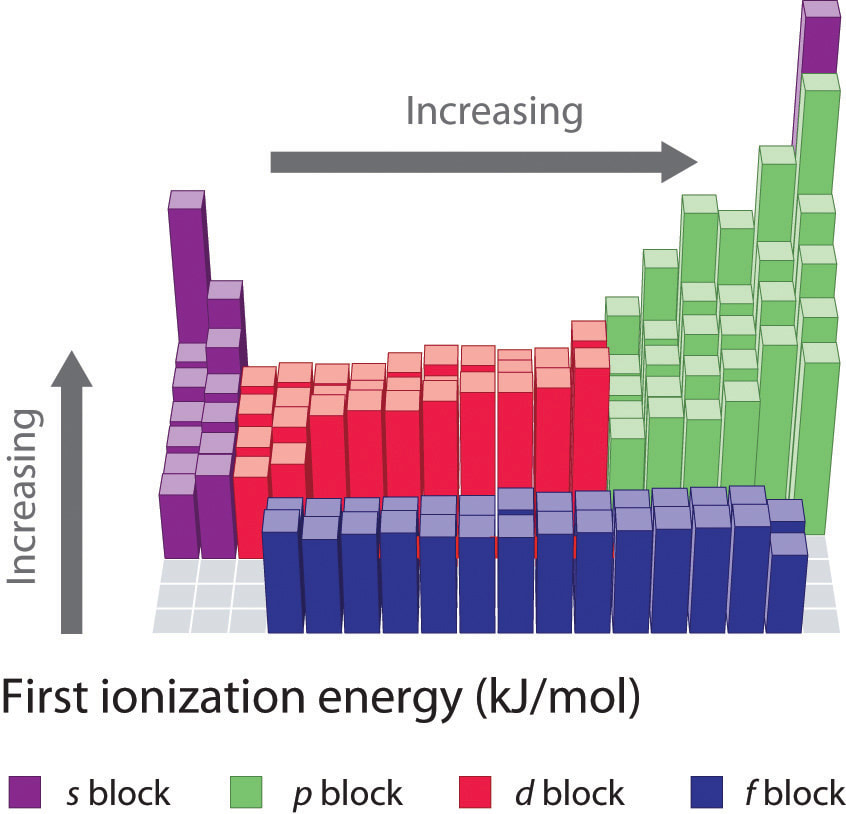
First Ionization Energies for the Elements of the Periodic Table.
| periodic-table-ions-fixed.pdf | |
| File Size: | 170 kb |
| File Type: | |
Metals
Carbon
Organic Compounds
In the older “trivial name” system of organic chemistry, the first four names in the hydrocarbon series are:--
methyl-, meth- (1 carbon atom)
The Germans created “Methyl” in the 1840s from the Ancient Greek words μέθυ (méthu, ‘wine’) and ὕλη (húlē, ‘wood’) to intentionally highlight the origins of methyl alcohol (wood alcohol) — being “alcohol made from wood (or woody substance).”
British English retains the original German pronunciation in methyl- as “mĕʹthĭl” (mee-theil).
American pronunciation is /ˈmɛθaɪl/ (meth-eil).
ethyl-, eth- (2 carbons)
The Germans also created the prefix from the German word Ether (sometimes Äther, ether) from the Latin aether (‘the upper pure bright air’) — in turn from Ancient Greek αἰθήρ (aithḗr, ‘upper air’).
However, the English word ether was from Old French ether.
The British English pronunciation is “ee-theil” (/ˈɛθaɪl/).
The American pronunciation is “ethel” (/ˈɛθəl/)
propyl-, prop- (3 carbons)
The French created the prefix from propane and from proprionic acid — whose French form acide proprionique was a coinage from the Ancient Greek πρῶτος (prôtos, ‘first’) and πίων (píōn, ‘fat’).
That’s because proprionic acid is the smallest carboxylic acid that exhibits the properties of fatty acids (i.e. producing an oily layer when salted out of water, and having a soapy potassium salt).
butyl-, but- (4 carbons)
An English prefix created in 1850s from butyric acid — from the Latin butyrum (‘butter’) — because butyric acid is a colourless, syrupy liquid found in rancid butter. Ultimately from Ancient Greek βούτυρος (boúturos, “butter”).
The remaining prefixes based on Greek numbers for the number of carbon atoms in the chain:--
5 carbons — pentyl-, pent- (e.g. pentane)
6 carbons — hexyl-, hex- (hexane)
7 carbons — heptyl-, hept- (heptane)
8 carbons — octyl-, oct- (octane)
9 carbons — nonyl-, nona- (nonane)
10 carbons — decyl, dec- (decane) — ‘c’ pronounced as “k”
11 carbons — undecyl-, undec- (undecane)
12 carbons — dodecyl-, dodec- (dodecane)
13 carbons — tridecyl-, tridec- (tridecane)
methyl-, meth- (1 carbon atom)
The Germans created “Methyl” in the 1840s from the Ancient Greek words μέθυ (méthu, ‘wine’) and ὕλη (húlē, ‘wood’) to intentionally highlight the origins of methyl alcohol (wood alcohol) — being “alcohol made from wood (or woody substance).”
British English retains the original German pronunciation in methyl- as “mĕʹthĭl” (mee-theil).
American pronunciation is /ˈmɛθaɪl/ (meth-eil).
ethyl-, eth- (2 carbons)
The Germans also created the prefix from the German word Ether (sometimes Äther, ether) from the Latin aether (‘the upper pure bright air’) — in turn from Ancient Greek αἰθήρ (aithḗr, ‘upper air’).
However, the English word ether was from Old French ether.
The British English pronunciation is “ee-theil” (/ˈɛθaɪl/).
The American pronunciation is “ethel” (/ˈɛθəl/)
propyl-, prop- (3 carbons)
The French created the prefix from propane and from proprionic acid — whose French form acide proprionique was a coinage from the Ancient Greek πρῶτος (prôtos, ‘first’) and πίων (píōn, ‘fat’).
That’s because proprionic acid is the smallest carboxylic acid that exhibits the properties of fatty acids (i.e. producing an oily layer when salted out of water, and having a soapy potassium salt).
butyl-, but- (4 carbons)
An English prefix created in 1850s from butyric acid — from the Latin butyrum (‘butter’) — because butyric acid is a colourless, syrupy liquid found in rancid butter. Ultimately from Ancient Greek βούτυρος (boúturos, “butter”).
The remaining prefixes based on Greek numbers for the number of carbon atoms in the chain:--
5 carbons — pentyl-, pent- (e.g. pentane)
6 carbons — hexyl-, hex- (hexane)
7 carbons — heptyl-, hept- (heptane)
8 carbons — octyl-, oct- (octane)
9 carbons — nonyl-, nona- (nonane)
10 carbons — decyl, dec- (decane) — ‘c’ pronounced as “k”
11 carbons — undecyl-, undec- (undecane)
12 carbons — dodecyl-, dodec- (dodecane)
13 carbons — tridecyl-, tridec- (tridecane)